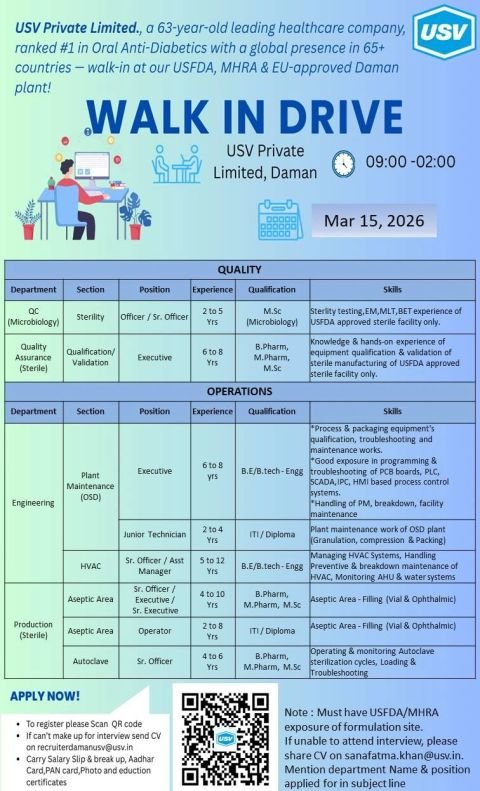
USV Private Limited, a leading Indian healthcare company and the #1 company in Oral Anti-Diabetics, has announced a Walk-in Drive for multiple pharma positions at its USFDA, MHRA, and EU-approved manufacturing facility in Daman.
The USV Pharma walk-in interview scheduled for 15 March 2026 is inviting experienced professionals from Quality Control, Quality Assurance, Production (Sterile), Engineering, HVAC, and Plant Maintenance departments. Candidates with USFDA or MHRA exposure in formulation manufacturing sites are strongly encouraged to attend.
This walk-in drive offers an excellent opportunity for pharma professionals seeking pharmaceutical manufacturing jobs in India, especially in USFDA-approved sterile manufacturing facilities.
Walk-in Interview Details
- Company: USV Private Limited
- Location: USV Manufacturing Plant, Daman
- Date: 15 March 2026
- Time: 09:00 AM – 02:00 PM
- Industry: Pharmaceutical Manufacturing
- Facility: USFDA, MHRA & EU approved formulation site
Available Pharma Job Openings
1. Quality Control – Microbiology (Sterility)
Position: Officer / Senior Officer
Experience: 2 – 5 Years
Qualification: M.Sc in Microbiology
Key Responsibilities
- Conduct sterility testing in sterile manufacturing environments
- Perform Environmental Monitoring (EM) and Microbial Limit Testing (MLT)
- Execute Bacterial Endotoxin Test (BET) procedures
- Maintain microbiology documentation as per regulatory requirements
- Ensure compliance with USFDA and MHRA regulatory guidelines
2. Quality Assurance – Sterile (Qualification & Validation)
Position: Executive
Experience: 6 – 8 Years
Qualification: B.Pharm / M.Pharm / M.Sc
Responsibilities
- Equipment qualification and validation of sterile manufacturing systems
- Review validation protocols and reports
- Ensure compliance with GMP and regulatory requirements
- Support regulatory audits and documentation review
3. Engineering – Plant Maintenance (OSD)
Position: Executive
Experience: 6 – 10 Years
Qualification: B.E / B.Tech (Engineering)
Responsibilities
- Troubleshooting of process and packaging equipment
- Programming and maintenance of PLC, SCADA, HMI, IPC systems
- PCB board troubleshooting and process automation support
- Preventive and breakdown maintenance of manufacturing equipment
4. Engineering – Junior Technician
Experience: 2 – 4 Years
Qualification: ITI / Diploma
Responsibilities
- Maintenance of OSD plant equipment including granulation, compression, and packing machines
- Support engineering team in breakdown maintenance
- Maintain equipment logs and documentation
5. HVAC – Sterile Manufacturing
Position: Senior Officer / Assistant Manager
Experience: 5 – 12 Years
Qualification: B.E / B.Tech Engineering
Responsibilities
- Manage HVAC systems in sterile manufacturing areas
- Preventive and breakdown maintenance of HVAC equipment
- Monitor AHU systems and purified water systems
- Ensure compliance with GMP environmental requirements
6. Production – Sterile (Aseptic Area)
Position: Sr Officer / Executive / Sr Executive
Experience: 4 – 10 Years
Qualification: B.Pharm / M.Pharm / M.Sc
Responsibilities
- Operate sterile filling lines for vials and ophthalmic products
- Maintain aseptic practices in sterile manufacturing areas
- Documentation and batch manufacturing record compliance
7. Production – Autoclave
Position: Senior Officer
Experience: 2 – 8 Years
Qualification: B.Pharm / M.Pharm / M.Sc
Responsibilities
- Operate and monitor autoclave sterilization cycles
- Perform loading and unloading of sterilization equipment
- Troubleshoot sterilization process issues
8. Production – Aseptic Area Operator
Experience: 4 – 6 Years
Qualification: ITI / Diploma
Responsibilities
- Operate filling machines in sterile areas
- Follow aseptic practices and SOPs
- Maintain batch documentation and production logs
Required Skills
Candidates applying for these pharma manufacturing jobs in India should have:
- Experience in USFDA / MHRA regulated pharmaceutical facilities
- Knowledge of GMP, sterile manufacturing, and validation
- Exposure to pharmaceutical production, QC microbiology, engineering maintenance, or HVAC systems
- Strong documentation and compliance knowledge
Benefits of Working at USV Pharma
Working with USV Private Limited offers several advantages:
- Opportunity to work in USFDA-approved sterile manufacturing plants
- Exposure to global pharmaceutical regulatory standards
- Career growth in quality assurance, production, and engineering roles
- Competitive salary packages and professional development
- Work with one of India’s leading anti-diabetic pharmaceutical companies
Documents Required for Walk-in Interview
Candidates attending the USV walk-in drive in Daman should carry:
- Updated Resume / CV
- Salary Slip and Salary Breakup
- Aadhaar Card
- PAN Card
- Passport-size Photographs
- Educational Certificates
How to Apply
Candidates who cannot attend the walk-in interview can send their CV via email.
Email:
recruiterdamanusv@usv.in
sanafatma.khan@usv.in