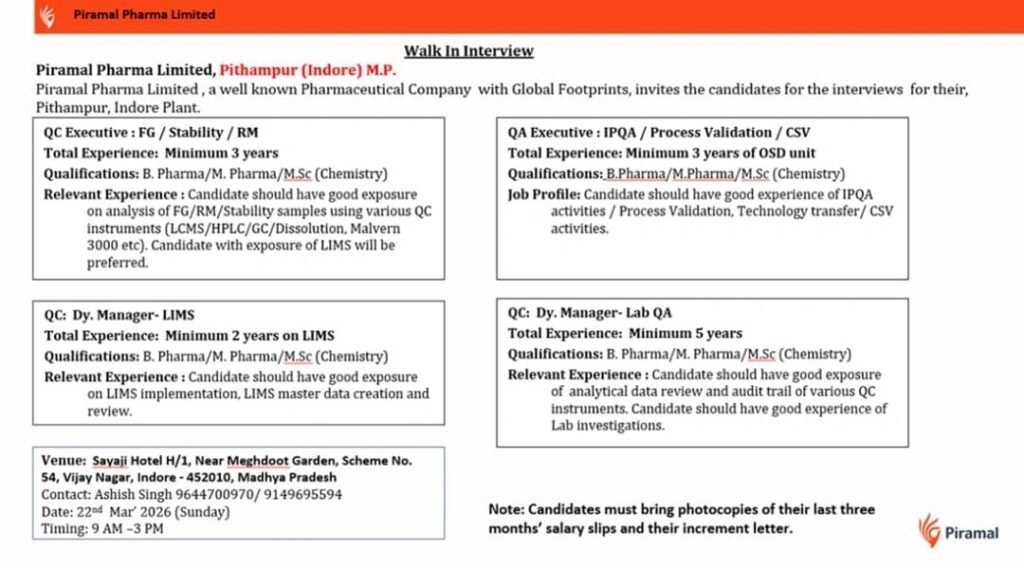
Piramal Pharma Limited, a globally recognized pharmaceutical company, is conducting a walk-in interview in Indore (Pithampur plant) for multiple QC and QA positions. This is an excellent opportunity for candidates with experience in Quality Control (QC), Quality Assurance (QA), and LIMS to join a reputed pharma organization with global operations.
If you are searching for pharma jobs in Indore, QC Executive jobs, QA Executive pharma jobs, or LIMS jobs in pharma, this walk-in drive could be the right opportunity.
📍 Walk-In Interview Details
- Company: Piramal Pharma Limited
- Location: Pithampur (Indore), Madhya Pradesh
- Interview Date: 22nd March 2026 (Sunday)
- Time: 9:00 AM – 3:00 PM
- Venue: Sayaji Hotel, H/1, Near Meghdoot Garden, Scheme No. 54, Vijay Nagar, Indore – 452010
- Contact: 9644700970 / 9149695594
💼 Job Openings
1. QC Executive – FG / Stability / RM
Experience: Minimum 3 years
Qualification: B.Pharm / M.Pharm / M.Sc (Chemistry)
Key Responsibilities:
- Analysis of Finished Goods (FG), Raw Materials (RM), and Stability samples
- Handling instruments like HPLC, GC, LCMS, Dissolution, Malvern 3000
- Ensuring compliance with QC procedures
- Exposure to LIMS systems preferred
2. QC Dy. Manager – LIMS
Experience: Minimum 2 years in LIMS
Qualification: B.Pharm / M.Pharm / M.Sc (Chemistry)
Key Responsibilities:
- LIMS implementation and configuration
- Master data creation and review
- Ensuring smooth integration of laboratory systems
3. QA Executive – IPQA / Process Validation / CSV
Experience: Minimum 3 years (OSD unit required)
Qualification: B.Pharm / M.Pharm / M.Sc (Chemistry)
Key Responsibilities:
- In-process quality assurance (IPQA) activities
- Process validation and technology transfer
- Computer System Validation (CSV)
4. QC Dy. Manager – Lab QA
Experience: Minimum 5 years
Qualification: B.Pharm / M.Pharm / M.Sc (Chemistry)
Key Responsibilities:
- Analytical data review and audit trail verification
- Handling lab investigations and deviations
- Ensuring compliance with regulatory standards
🎓 Qualifications & Skills
- Degree in Pharmacy or Chemistry (B.Pharm / M.Pharm / M.Sc)
- Strong knowledge of QC instruments and QA processes
- Experience in regulated pharma environments (OSD preferred)
- Familiarity with LIMS, validation, and compliance systems
💰 Salary & Benefits
- Estimated Salary Range: ₹4,00,000 – ₹12,00,000 per annum (based on role & experience)
- Opportunity to work with a global pharmaceutical company
- Exposure to advanced QC instruments and regulatory systems
- Career growth in QA, QC, and LIMS domains
📄 Documents Required
Candidates must carry:
- Updated Resume
- Photocopies of last 3 months’ salary slips
- Latest increment letter
- Educational certificates