Cipla Limited, one of India’s most trusted pharmaceutical companies, is conducting a Walk-in Interview on 4 January 2026 (Sunday) for multiple API and Formulation Engineering & Production roles. This hiring drive is ideal for freshers (2025 pass-outs) as well as experienced professionals looking to build a long-term career in API manufacturing, formulation production, and engineering operations.
This opportunity is based at Cipla’s Kurkumbh manufacturing facility, Maharashtra, with the interview venue in Aurangabad (Chhatrapati Sambhajinagar).
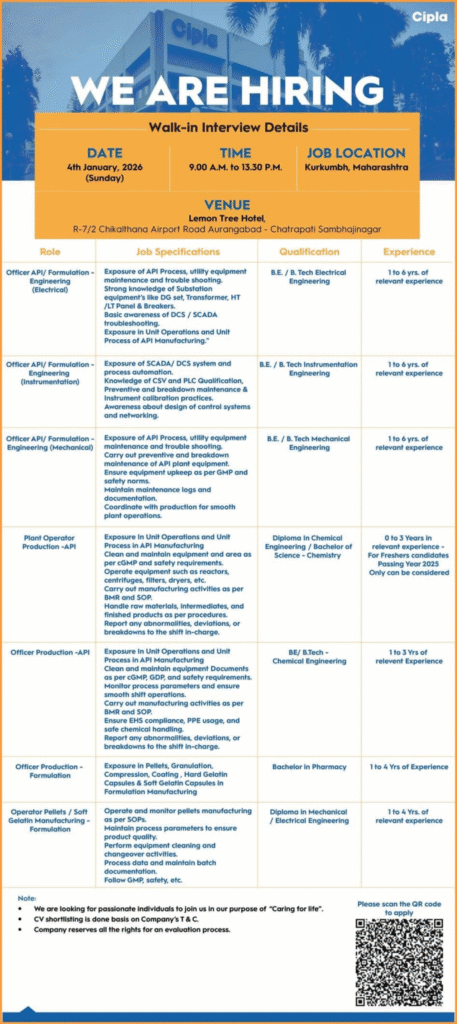
Cipla Walk-in Interview Details
- Company: Cipla Limited
- Date: 4 January 2026 (Sunday)
- Time: 9:00 AM to 1:30 PM
- Job Location: Kurkumbh, Maharashtra
- Interview Venue:
Lemon Tree Hotel,
R-7/2, Chikalthana Airport Road,
Aurangabad – Chhatrapati Sambhajinagar, Maharashtra
Open Positions at Cipla
Engineering – API / Formulation
- Officer – API/Formulation Engineering (Electrical)
- Officer – API/Formulation Engineering (Instrumentation)
- Officer – API/Formulation Engineering (Mechanical)
Production – API & Formulation
- Plant Operator – Production (API)
- Officer / In-Charge – Production (API)
- Officer – Production (Formulation)
- Operator – Pellets / Soft Gelatin / Formulation Manufacturing
Key Responsibilities
Engineering Roles
- Maintenance and troubleshooting of API process & utility equipment
- Handling DG sets, transformers, substations, HT/LT panels & breakers
- Preventive and breakdown maintenance as per GMP & safety norms
- Exposure to DCS/SCADA systems, PLC, CSV qualification
- Instrument calibration, documentation, and equipment upkeep
- Coordination with production teams for smooth plant operations
Production Roles (API & Formulation)
- Exposure to unit operations and API manufacturing processes
- Operation of reactors, centrifuges, dryers, filters, and granulation equipment
- Pelletization, compression, coating, HGC & soft gelatin capsule manufacturing
- BMR/BPR documentation, SOP adherence, and shift monitoring
- Compliance with cGMP, GDP, EHS, PPE usage, and chemical safety
- Reporting deviations, abnormalities, and breakdowns
Eligibility & Qualifications
| Qualification | Experience |
|---|---|
| BE/B.Tech – Electrical Engineering | 1–4 Years |
| BE/B.Tech – Instrumentation Engineering | 1–6 Years |
| BE/B.Tech – Mechanical Engineering | 1–6 Years |
| Diploma Chemical / B.Sc Chemistry | 0–3 Years (Freshers – 2025 pass-outs only) |
| BE/B.Tech – Chemical Engineering | 1–3 Years |
| B.Pharm | 1–4 Years |
| Diploma Mechanical / Electrical | 1–4 Years |
Why Join Cipla?
- Work with a globally respected pharmaceutical brand
- Strong exposure to API & formulation manufacturing
- Learning-oriented environment with structured SOPs & GMP systems
- Competitive salary aligned with industry standards
- Career growth opportunities across manufacturing and engineering functions
Cipla’s purpose of “Caring for Life” ensures employees grow while making a meaningful impact on global healthcare.