Senores Pharmaceuticals is a global pharmaceutical company focused on developing and manufacturing high-quality medicines for regulated markets like the US, Canada, and emerging markets. The company specializes in multiple dosage forms and therapeutic segments, ensuring global compliance and innovation.
Advertisement
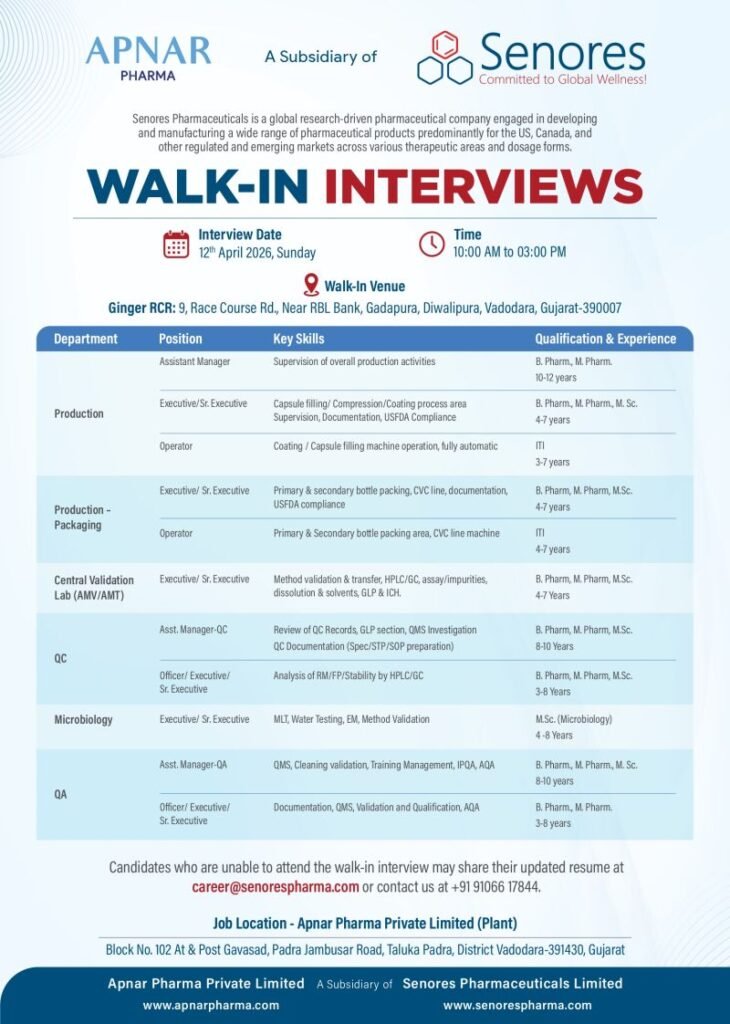
📅 Walk-In Interview Details
- Interview Date: 12th April 2026 (Sunday)
- Time: 10:00 AM – 03:00 PM
- Venue: Ginger RCR, Race Course Road, Vadodara, Gujarat
📍 Job Location: Apnar Pharma Pvt. Ltd. (Subsidiary of Senores Pharmaceuticals), Vadodara
💼 Open Positions & Departments
Production Department
- Assistant Manager (10–12 Years)
- Executive / Sr. Executive (4–7 Years)
- Operator (3–7 Years)
Production – Packaging
- Executive / Sr. Executive (4–7 Years)
- Operator (4–7 Years)
Quality Control (QC)
- Assistant Manager (8–10 Years)
- Officer / Executive / Sr. Executive (3–8 Years)
Quality Assurance (QA)
- Assistant Manager (8–10 Years)
- Officer / Executive / Sr. Executive (3–8 Years)
Microbiology
- Executive / Sr. Executive (4–8 Years)
Central Validation Lab
- Executive / Sr. Executive (4–7 Years)
🧪 Key Responsibilities
- Supervise pharmaceutical production & packaging operations
- Ensure USFDA compliance and documentation
- Perform method validation, HPLC/GC analysis, and QC testing
- Handle QMS, IPQA, audits, and investigation processes
- Execute microbiology testing (MLT, EM, water testing)
- Maintain GLP, ICH guidelines, and validation protocols
🎓 Qualifications Required
- B.Pharm / M.Pharm / M.Sc (Chemistry / Microbiology)
- Relevant experience in pharmaceutical manufacturing or quality systems
- Strong knowledge of USFDA, GLP, QMS, Validation, Documentation
💰 Salary & Benefits
- Estimated Salary Range: ₹3.5 LPA – ₹14 LPA (based on role & experience)
- Work with a global pharma company
- Exposure to regulated markets (USFDA)
- Career growth in quality, production & validation domains
- Professional and compliant work environment
📩 Application Process
👉 Walk-in directly at the venue with:
- Updated Resume
- Educational Certificates
- Experience Documents
📧 If unable to attend, send your CV to: career@senorespharma.com
Advertisement