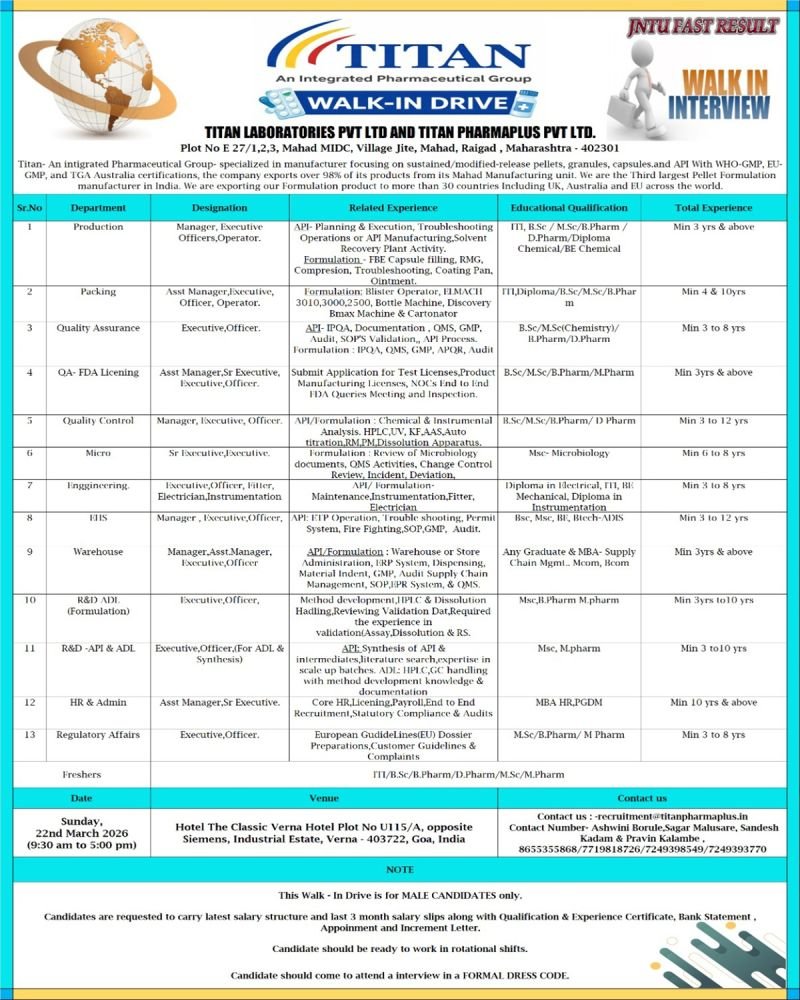
Titan Laboratories Pvt. Ltd. and Titan Pharmaplus Pvt. Ltd., a globally recognized integrated pharmaceutical group, are conducting a walk-in interview drive for multiple pharmaceutical roles. The company is inviting experienced professionals from Production, Quality Assurance, Quality Control, R&D, Engineering, Regulatory Affairs, Warehouse, HR, and Packing departments.
The Titan Pharma Walk-In Drive 2026 will take place on 22 March 2026 in Goa, offering excellent opportunities for professionals with 3–12 years of pharmaceutical industry experience.
Titan Pharma is known for its WHO-GMP, EU-GMP, and TGA Australia certified manufacturing facilities and exports over 98% of its pharmaceutical formulations and APIs to more than 30 countries, including the UK, EU, and Australia. The company is also recognized as the third-largest pellet formulation manufacturer in India.
Walk-In Interview Details
- Company: Titan Laboratories Pvt. Ltd. & Titan Pharmaplus Pvt. Ltd.
- Interview Date: Sunday, 22 March 2026
- Time: 9:30 AM – 5:00 PM
- Venue: Hotel The Classic Verna Hotel, Plot No U115/A, Opposite Siemens, Industrial Estate, Verna, Goa – 403722
- Work Location: Mahad MIDC, Raigad, Maharashtra
- Eligibility: Male candidates only
- Shift: Rotational shifts
Available Departments & Roles
Titan Pharma is hiring across multiple pharmaceutical departments.
Production Department
Designations: Manager, Executive, Officer, Operator
Key Responsibilities
- API manufacturing operations and troubleshooting
- Recovery plant activities and solvent recovery
- Handling pellet formulations, capsule filling, compression, coating pan, RMG operations
- Ointment and formulation manufacturing activities
Experience: Minimum 3+ years
Packing Department
Designations: Assistant Manager, Executive, Officer, Operator
Responsibilities
- Blister packing operations
- Handling machines such as ELMACH 3010/3000/2500
- Bottle packing and cartonator operations
- Ensuring GMP compliance during packaging
Experience: 4–10 years
Quality Assurance (QA)
Designations: Assistant Manager, Senior Executive, Executive, Officer
Responsibilities
- API/Formulation IPQA activities
- Documentation, QMS, OMS, SOP compliance
- Validation and audit management
- Handling regulatory inspections and FDA licensing documentation
Experience: 3–8 years
Quality Control (QC)
Designations: Manager, Executive, Officer
Responsibilities
- Chemical and instrumental analysis
- Handling instruments such as HPLC, UV, KF, AAS, auto titration systems
- Dissolution testing for RM and PM samples
- Data analysis and documentation
Experience: 3–12 years
Microbiology Department
Designations: Senior Executive, Executive
Responsibilities
- Microbiology documentation review
- Change control and deviation management
- Environmental monitoring and microbiological analysis
Qualification: MSc Microbiology
Experience: 6–8 years
Engineering Department
Designations: Executive, Officer, Fitter, Electrician, Instrumentation
Responsibilities
- Maintenance and engineering support for API and formulation plants
- Electrical, mechanical and instrumentation maintenance
Qualification: Diploma / ITI / Engineering
Experience: 3–8 years
EHS Department
Responsibilities
- ETP operation and environmental monitoring
- Permit system management
- Fire safety compliance
- GMP and audit support
Experience: 3–12 years
Warehouse / Supply Chain
Responsibilities
- Warehouse and store administration
- ERP and inventory management
- Material dispensing and supply chain operations
- GMP documentation
R&D – Formulation (ADE)
Responsibilities
- Method development and validation
- Handling HPLC and dissolution systems
- Validation of assay and related substances
Experience: 3–10 years
R&D – API / ADL
Responsibilities
- API synthesis and scale-up batches
- Literature search and intermediate synthesis
- Analytical development using HPLC and GC
HR & Admin
Responsibilities
- Core HR operations
- Payroll processing and statutory compliance
- Recruitment and HR audits
Regulatory Affairs
Responsibilities
- EU dossier preparation
- Handling regulatory documentation
- Customer queries and regulatory compliance
Educational Qualifications
Eligible candidates should possess any of the following qualifications:
- BSc / MSc (Chemistry, Microbiology)
- B.Pharm / M.Pharm
- D.Pharm
- Diploma Chemical Engineering
- BE / B.Tech Chemical
- ITI / Diploma Electrical / Mechanical / Instrumentation
- MBA / PGDM HR or Supply Chain
Benefits of Working with Titan Pharma
- Opportunity to work in a WHO-GMP and EU-GMP certified pharmaceutical company
- Exposure to global pharmaceutical markets across 30+ countries
- Work with one of India’s leading pellet formulation manufacturers
- Career growth opportunities in API and formulation manufacturing
- Competitive salary packages and professional development
Documents Required for Walk-In Interview
Candidates must bring:
- Updated Resume
- Educational Certificates
- Experience Certificates
- Latest Salary Structure
- Last 3 months Salary Slips
- Bank Statement
- Appointment & Increment Letters
Dress Code: Formal attire
Contact Information
For queries regarding the Titan Pharma Walk-In Drive: