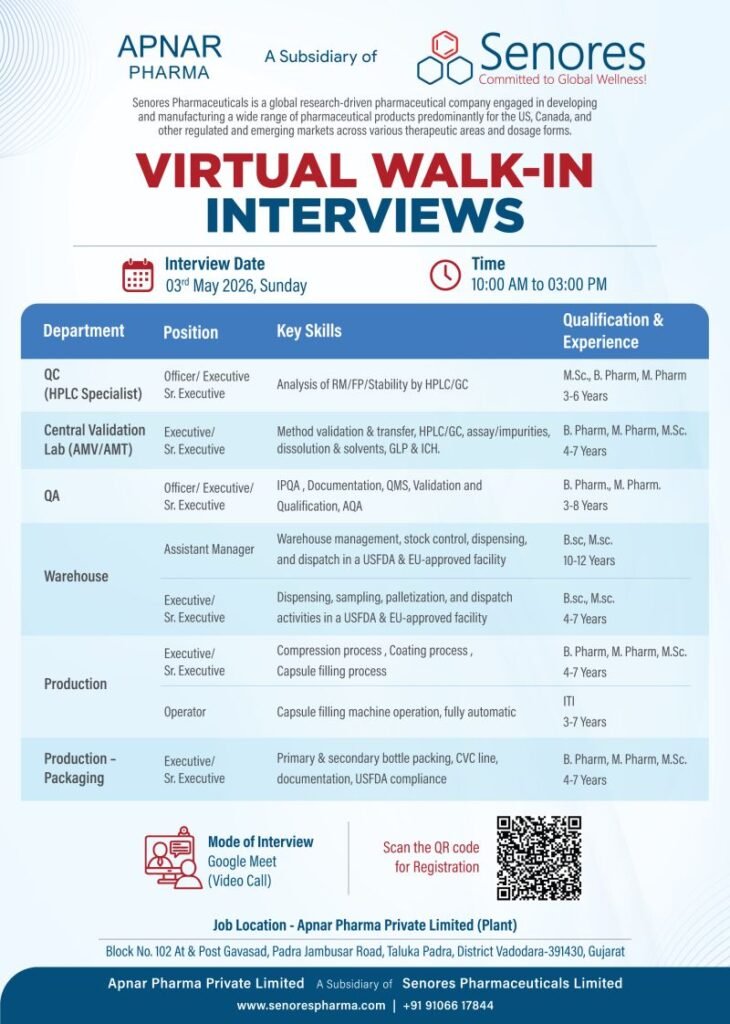
Apnar Pharma Private Limited, a subsidiary of Senores Pharmaceuticals, is conducting a virtual walk-in interview on 3rd May 2026 for multiple positions in QC, QA, Production, Warehouse, and Validation departments.
This is a great opportunity for experienced pharma professionals seeking jobs in a USFDA-approved facility with global exposure in regulated markets like the US and Canada.
📅 Interview Details
- Interview Date: 03 May 2026 (Sunday)
- Time: 10:00 AM – 03:00 PM
- Mode: Virtual (Google Meet Video Call)
- Job Location: Vadodara, Gujarat
💼 Available Job Openings
1. QC (HPLC Specialist)
Position: Officer / Executive / Sr. Executive
Key Skills:
- HPLC/GC analysis
- Raw Material (RM), Finished Product (FP), Stability testing
Experience: 3–6 Years
Qualification: M.Sc / B.Pharm / M.Pharm
2. Central Validation Lab (AMV/AMT)
Position: Executive / Sr. Executive
Key Skills:
- Method validation & transfer
- HPLC/GC, assay, impurities, dissolution
- GLP & ICH guidelines
Experience: 4–7 Years
Qualification: B.Pharm / M.Pharm / M.Sc
3. Quality Assurance (QA)
Position: Officer / Executive / Sr. Executive
Key Skills:
- IPQA, Documentation
- QMS, Validation & Qualification
- AQA
Experience: 3–8 Years
Qualification: B.Pharm / M.Pharm
4. Warehouse – Assistant Manager
Key Skills:
- Warehouse management
- Stock control, dispensing, dispatch
- USFDA & EU regulatory compliance
Experience: 10–12 Years
Qualification: B.Sc / M.Sc
5. Warehouse Executive
Position: Executive / Sr. Executive
Key Skills:
- Dispensing, sampling
- Palletization & dispatch
Experience: 4–7 Years
Qualification: B.Sc / M.Sc
6. Production (Manufacturing)
Position: Executive / Sr. Executive
Key Skills:
- Compression, coating processes
- Capsule filling
Experience: 4–7 Years
Qualification: B.Pharm / M.Pharm / M.Sc
7. Production Operator
Key Skills:
- Capsule filling machine operation (fully automatic)
Experience: 3–7 Years
8. Production – Packaging
Position: Executive / Sr. Executive
Key Skills:
- Bottle packing (primary & secondary)
- CVC line operations
- USFDA documentation
Experience: 4–7 Years
Qualification: B.Pharm / M.Pharm / M.Sc
🎯 Key Responsibilities
- Perform analytical testing using HPLC/GC
- Ensure compliance with USFDA, ICH, and GLP guidelines
- Handle documentation, validation, and QMS processes
- Manage production and packaging operations
- Maintain warehouse inventory and dispatch processes
🎓 Required Qualifications
- B.Pharm / M.Pharm / M.Sc / B.Sc
- Relevant pharma industry experience (3–12 years)
- Strong knowledge of regulatory compliance and documentation
💰 Salary & Benefits
- Estimated Salary: ₹3.5 LPA – ₹12 LPA (based on role & experience)
- Opportunity to work in a USFDA-approved facility
- Exposure to regulated international markets
- Career growth in a global pharmaceutical company
📩 How to Apply