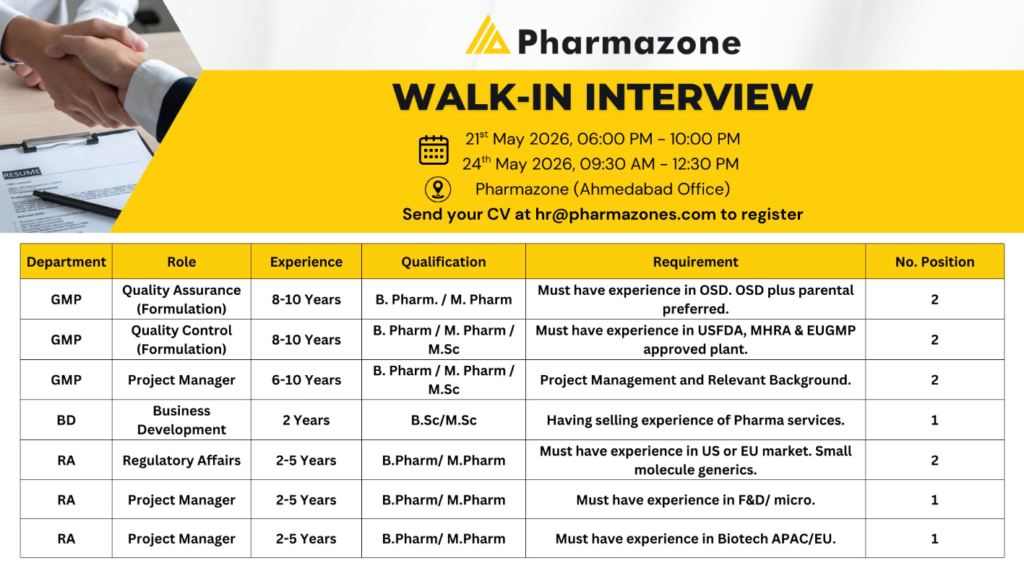
Pharmazone has announced a major walk-in interview drive for experienced professionals in GMP, Regulatory Affairs (RA), and Business Development (BD). Candidates with expertise in QA, QC, OSD formulations, regulatory submissions, project management, biotech, and pharma services can attend the walk-in interview on 21st May 2026 and 24th May 2026 at the Ahmedabad office.
This Pharmazone walk-in drive offers excellent career opportunities for B.Pharm, M.Pharm, M.Sc, and Life Sciences candidates seeking pharma jobs in Ahmedabad with exposure to USFDA, MHRA, EU-GMP, APAC, and biotech markets.
About Pharmazone
Pharmazone is a leading global life-sciences consulting partner known for delivering high-quality services in:
- Regulatory Affairs
- Pharmacovigilance
- GxP Compliance
- GMP Consulting
- Project Management
- Business Development Solutions
The company supports pharmaceutical and biotechnology organizations worldwide to accelerate innovation while ensuring regulatory compliance and patient safety.
Walk-In Interview Details
Interview Dates & Timings
- 21st May 2026 (Thursday) — 06:00 PM to 10:00 PM
- 24th May 2026 (Sunday) — 09:30 AM to 12:30 PM
Walk-In Venue
Pharmazone Ahmedabad Office
Ahmedabad, Gujarat, India
Registration
Interested candidates should send their updated CV to:
Available Job Openings at Pharmazone
| Department | Role | Experience | Qualification | Vacancies |
|---|---|---|---|---|
| GMP | Quality Assurance (Formulation) | 8–10 Years | B.Pharm / M.Pharm | 2 |
| GMP | Quality Control (Formulation) | 8–10 Years | M.Sc / B.Pharm / M.Pharm | 2 |
| GMP | Project Manager | 6–10 Years | B.Pharm / M.Pharm / M.Sc | 2 |
| Business Development | BD Executive | 2 Years | B.Sc / M.Sc | 1 |
| Regulatory Affairs | Regulatory Affairs Executive | 2–5 Years | B.Pharm / M.Pharm | 2 |
| Regulatory Affairs | Project Manager (F&D/Micro) | 2–5 Years | B.Pharm / M.Pharm | 1 |
| Regulatory Affairs | Project Manager (Biotech APAC/EU) | 2–5 Years | B.Pharm / M.Pharm | 1 |
Key Responsibilities
GMP – Quality Assurance & Quality Control
- Handle GMP documentation and compliance activities
- Ensure adherence to USFDA, MHRA, and EU-GMP guidelines
- Manage OSD formulation quality systems
- Conduct quality investigations and audits
- Support validation and regulatory inspections
Regulatory Affairs Roles
- Prepare and review regulatory dossiers
- Handle submissions for US, EU, APAC, and global markets
- Manage lifecycle maintenance activities
- Coordinate with cross-functional pharma teams
- Support product registration and approvals
Business Development
- Promote pharmaceutical consulting services
- Build relationships with pharma and biotech clients
- Generate leads and manage client communications
- Support business growth initiatives
Project Management
- Lead pharma and biotech consulting projects
- Coordinate timelines, deliverables, and compliance requirements
- Collaborate with regulatory, QA, QC, and development teams
Qualifications Required
Candidates applying for Pharmazone pharma jobs should possess:
- B.Pharm
- M.Pharm
- M.Sc
- B.Sc (for Business Development role)
Preferred Skills
- Experience in OSD formulations
- Knowledge of USFDA, MHRA, and EU-GMP regulations
- Regulatory Affairs expertise in US/EU markets
- Biotech APAC/EU regulatory exposure
- Strong communication and project management skills
Why Join Pharmazone?
Working with Pharmazone offers:
- Exposure to international pharma regulatory markets
- Career growth in GMP and Regulatory Affairs
- Opportunity to work with biotech and pharma leaders
- Professional development in life-sciences consulting
- Dynamic and collaborative work environment
How to Apply
Interested and eligible candidates can attend the walk-in interview directly on the scheduled dates.
Important Instructions
- Carry updated resume/CV
- Bring educational and experience documents
- Carry government photo ID proof
- Prior registration via email is preferred
📩 Send CV to: hr@pharmazones.com