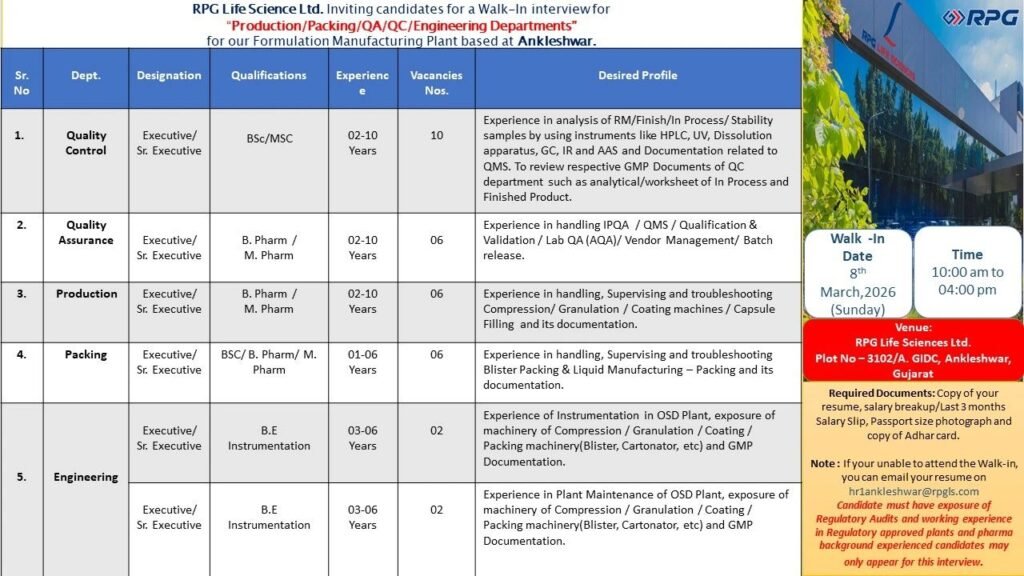
RPG Life Sciences Ltd. is inviting eligible pharma professionals for a Walk-In Interview on 8th March 2026 (Sunday) for multiple vacancies in Quality Control (QC), Quality Assurance (QA), Production, Packing, and Engineering Departments at its Formulation Manufacturing Plant in Ankleshwar, Gujarat.
This is an excellent opportunity for candidates with experience in regulatory-approved pharmaceutical plants, particularly those with exposure to Regulatory Audits, GMP documentation, OSD manufacturing, and QMS systems.
📅 Walk-In Interview Details
- Date: 8th March 2026 (Sunday)
- Time: 10:00 AM to 04:00 PM
- Venue: Plot No-3102/A, GIDC, Ankleshwar, Gujarat
- Email (If unable to attend): hriankleshwar@rpgls.com
🔎 Department-wise Vacancies & Eligibility
1️⃣ Quality Control (QC) – Executive/Sr. Executive
- Qualification: BSc / MSc
- Experience: 2–10 Years
- Vacancies: 10
Key Responsibilities:
- Analysis of RM, In-Process, Finished & Stability samples
- Hands-on experience with HPLC, UV, Dissolution, GC, IR, AAS
- QMS documentation and GMP compliance
- Review of analytical worksheets and finished product documentation
2️⃣ Quality Assurance (QA) – Executive/Sr. Executive
- Qualification: B.Pharm / M.Pharm
- Experience: 2–10 Years
- Vacancies: 6
Key Responsibilities:
- Handling IPQA, QMS, Qualification & Validation, Lab QA (AQA)
- Vendor management and batch release
- Exposure to regulatory audits
- GMP documentation review
3️⃣ Production – Executive/Sr. Executive
- Qualification: B.Pharm / M.Pharm
- Experience: 2–10 Years
- Vacancies: 6
Key Responsibilities:
- Supervising and troubleshooting:
- Compression
- Granulation
- Coating
- Capsule Filling
- Batch documentation and GMP compliance
- Experience in OSD plant operations
4️⃣ Packing – Executive/Sr. Executive
- Qualification: BSc / B.Pharm / M.Pharm
- Experience: 1–6 Years
- Vacancies: 6
Key Responsibilities:
- Blister Packing operations
- Liquid manufacturing – packing supervision
- Handling cartonators and packing line equipment
- Documentation as per GMP norms
5️⃣ Engineering – Executive/Sr. Executive
- Qualification: B.E Instrumentation
- Experience: 3–6 Years
- Vacancies: 2
Key Responsibilities:
- Instrumentation in OSD Plant
- Maintenance of Compression, Granulation, Coating & Packing machinery
- Exposure to Blister & Cartonator machines
- GMP documentation & regulatory compliance
🎯 Desired Candidate Profile
- Experience in Regulatory Approved Pharmaceutical Plants
- Exposure to USFDA/MHRA/WHO regulatory audits
- Strong understanding of GMP documentation & QMS
- Pharma background candidates only
📄 Required Documents for Walk-In
- Updated Resume (Hard Copy)
- Last 3 Months Salary Slips / Salary Breakup
- Passport Size Photograph
- Copy of Aadhaar Card
💼 Why Join RPG Life Sciences?
- Work in a regulatory-approved formulation manufacturing plant
- Exposure to global compliance standards
- Career growth in QA, QC, Production & Engineering
- Competitive salary as per industry standards
- Opportunity to work with experienced pharma professionals
📌 How to Apply?
Interested candidates can:
- Attend the Walk-In Interview on 8 March 2026
- Or email their resume to hriankleshwar@rpgls.com if unable to attend